There have been significant developments in health policy over the past few weeks. I highlight several of those below, including misguided Biden administration rules restricting laboratory-developed tests and Association Health Plans and two former secretaries of the Department of Health and Human Services who represent different political parties but agree on Medicare site neutral payments. But, first, Paragon released a new report today from Joe Albanese, Reducing Government Subsidies for Wealthier Medicare Enrollees.
Reducing Government Subsidies for Wealthier Medicare Enrollees
What this paper covers and what we found
- The paper examines current means-testing policies in Medicare and potential approaches for expanding it.
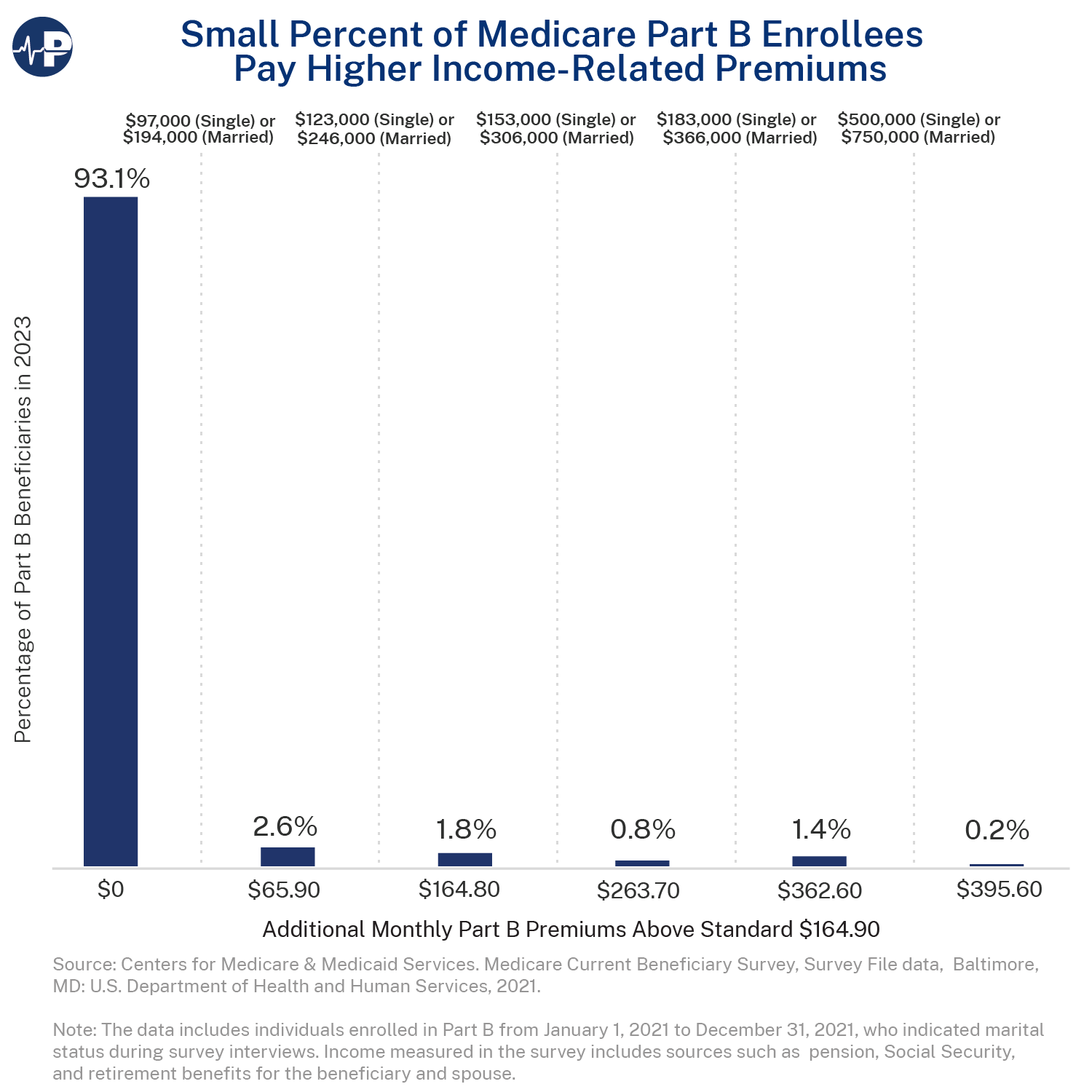
- For 93 percent of enrollees in 2023, taxpayers picked up about 75 percent of the cost of Part B, which covers outpatient and physician services. Means-testing in Medicare refers to higher-income enrollees paying additional premiums. In 2023, these higher premiums applied to enrollees with annual incomes of at least $97,000 ($194,000 for married couples) two years prior.
- As today’s Paragon Pic demonstrates, the current scope of means-testing in Medicare is quite limited. Higher Part B premiums applied to only about 7 percent of Part B beneficiaries in 2023. The Pic shows the additional premium payments beyond the standard monthly amount, i.e., the reduction in taxpayer subsidies, for higher-income Part B enrollees, and the percentage of enrollees who fall into each of the six premium tiers based on their annual household income.
Why it matters
- Medicare represents the nation’s primary fiscal challenge, and given rising federal debt and interest payments, building on existing but limited means-testing policies for wealthier beneficiaries could help shore up the program’s finances and improve the country’s budgetary outlook.
- Despite current means-testing policies, wealthier Medicare enrollees currently have 15 to 65 percent of their estimated Part B costs subsidized by taxpayers, and enrollees across income levels receive far more in benefits than they pay in payroll taxes during their working years and in premiums during retirement.
Policy Suggestions
- Policymakers should reduce government subsidies for wealthier beneficiaries. This can be accomplished a few different ways, including by (1) adjusting the income thresholds at which means-testing applies or the additional premium amounts paid under current law (as proposed by numerous organizations in the past), (2) expanding the measures of wealth, including lifetime income, that are used to target means-testing policies, and (3) considering alternative mechanisms for means-testing besides beneficiary premiums.
Misguided and Likely Unlawful FDA Rule Restricting LDTs
The Food and Drug Administration has issued a final rule that explicitly asserts its claim of authority to regulate laboratory-developed tests (LDTs) as medical devices. LDTs are tests that are designed, manufactured, and used with a single laboratory that is certified to perform high-complexity testing. As explained in a Paragon paper authored by Dr. Joel Zinberg, this rule will impose costly and time-consuming FDA regulation on LDTs which will curb innovation that is important to combatting emerging infectious diseases and cancer. Joel also explained how the FDA likely lacks statutory authority to regulate LDTs.
Under the Supreme Court’s major questions doctrine, an agency may not presume it has the authority to regulate an issue of great “economic and political significance” unless it can “point to ‘clear congressional authorization’ for the power it claims.” The Food, Drug, and Cosmetic Act (FDCA) does not mention regulating laboratories or laboratory testing services and various sections of the statute suggest it does not apply to LDTs. Moreover, FDA has never regulated LDTs, citing “enforcement discretion.” The Supreme Court noted in a 2014 case, “When an agency claims to discover in a long-extant statue an unheralded power to regulate ‘a significant portion of the American economy’ we typically greet its announcement with a measure of skepticism.”
FDA has clear authority to regulate devices developed for commercial distribution to many laboratories. LDTs, though, are methodologies and processes developed and used in a single laboratory to generate results for medical practitioners. They are more like medical services which the FDCA specifically bars the FDA from regulating.
This rule will undoubtedly face legal challenges as it should. Changes to FDA’s regulatory authority should come from Congress, not from bureaucrats.
Restricting Options for Small Employers
The Department of Labor has issued a rule rescinding its 2018 rule that had expanded the ability of employers to offer their workers coverage through an Association Health Plan (AHP). AHPs enable employers to offer high-quality coverage that avoids some costly government regulatory requirements and permits employers to pool their workers to take advantage of economies of scale and improved bargaining power with providers. (For more on AHPs, see Paragon’s policy brief, Small Business Health Insurance Equity Through Association Health Plans.)
As we explained in a comment letter joined by over a dozen policy experts, this rule harms small employers and their workers and is based on a faulty analysis. Policymakers should be looking to expand options for small businesses and workers, not take them away.
Secretaries Azar and Sebelius Support Medicare Site Neutral Payments
Two former Department of Health & Human Services secretaries on opposite sides of the political spectrum recently published a column where they agreed that enacting site-neutral payment policy is a crucial step to reduce health care costs for patients. Secretaries Alex Azar and Kathleen Sebelius compare a hospital buying up an independent clinic to a global coffee chain buying an independent shop, and doubling the price of a cup of coffee overnight – the customer pays drastically higher prices for nothing more than a change in ownership. The former secretaries also cite MedPAC’s 2022 Report to Congress, which shows that Medicare paid 141% more for cancer patient care at a hospital clinic compared to care at a freestanding office.
While the Obama and Trump administrations both supported site-neutrality and pursued means of reform (unlike the Biden administration), more work remains to end hospital overpayments and save money for seniors and taxpayers. A Paragon policy brief from last year analyzed several ways to enact site neutral policies, which can be a crucial tool for improving Medicare’s finances.
All the best,
Brian Blase
President
Paragon Health Institute



